UTAS Home › › Mathematics Pathways › Pathways to Health Science › Module 4: Measurement, units and chemistry calculations for Health Science
Click on the link below to take the Pre-Test for Module 4. The Pre-Test is optional but we recommend taking it to test your knowledge of Measurement, Units and Chemistry Calculations for Health Science. There are only 5 questions and it will only take about 10 minutes to complete.
If you receive 80% or greater on the Pre-Test, you have a good knowledge of Measurement, Units and Chemistry Calculations for Health Science and can move on to the next module or review the materials in module 4.
If you receive less than an 80%, work your way through the module and then take the quiz at the end to test your knowledge.
Before we look more closely at some specific chemistry calculations, you may find it valuable to review some of the key ideas in relation to the mathematics you will be dealing with.
Ratio is a way of comparing the size
of one quantity with the size of another. Ratios are expressed in the form m
: n or ![]() (n ≠ 0) and the comparison of two quantities may or
may not refer to the same attribute. For example, a volume may be compared to
another volume as in 60 ml to 45 ml, or a volume may be compared to time as in
60 ml per minute (i.e., 60 ml/minute). Please click on the following link to Math Is Fun - Maths Resources
for further explanation and examples. You will also find links to
several practice questions towards the bottom of the webpage.
(n ≠ 0) and the comparison of two quantities may or
may not refer to the same attribute. For example, a volume may be compared to
another volume as in 60 ml to 45 ml, or a volume may be compared to time as in
60 ml per minute (i.e., 60 ml/minute). Please click on the following link to Math Is Fun - Maths Resources
for further explanation and examples. You will also find links to
several practice questions towards the bottom of the webpage.
Proportional reasoning involves accounting for the relative amounts of two or more multiplicatively related quantities. For example the ratios 2:3 and 6:9 are said to be in proportion because they are equal. That is 2 : 3 = (2 x 3) : (3 x 3) = 6 : 9. Please click on the following link to Math Is Fun - Maths Resources for further explanation and examples. You will also find links to several practice questions towards the bottom of the webpage.
Recall that percentages are a way of thinking about any collection or quantity in terms of 100 parts and this enables us to describe and compare different quantities.
Example 1
We can compare the instance of a particular disease in different populations by comparing the relative proportions of those affected by the disease.
60.3 percent of men, and 66.6 percent of women aged 18 years and over have a waist circumference that put them at an increased risk of developing chronic disease.
Percent means per one hundred.
Example 2
If there are 200 people living in a small village, and 50 of these are children, this could be expressed as 25 children per 100 people or 25% (recall also that 25% can be written as the decimal 0.25 or as the fraction ![]() ).
).
We can also express this as a ratio.
That is, if 25% of our small population are children we can say that one in every four people is a child so this means that the ratio of children to adults in that population is one to three (1:3).
The following web link from Math Goodies deals with calculations involving percentages, including how to find a percentage of a quantity, and how to express one quantity as a percentage of another. Please click on the link and work through the explanations and the exercises provided.
The metric system of measurement is based upon powers of 10.
"Powers of 10" are created by multiplying by tens. For example:
10 x 10 = 100
10 x 10 x 10 = 1 000
10 x 10 x 10 x 10 = 10 000
10 x 10 x 10 x 10 x 10 = 100 000 (ten times ten times ten times ten times ten
equals one hundred thousand).
Powers of 10 can be written using exponents, for example, 103 = 1
000. The "3" indicates that 3 tens have been multiplied together. There are
also 3 zeros in the product (i.e., 10 x 10 x 10 = 1000).
Powers of 10 with an exponent of 1 or greater are counting numbers {1, 2, 3, 4, ...}.
Notice that in the table below, moving one column to the right equates to multiplication by 10 and moving one column to the left equates to division by 10. It is also important to recognise the equivalent ways of representing powers of 10.
Example
10-3 = ![]() = 0.001
= 0.001
The following Table shows the powers of 10.
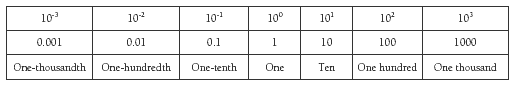
The following table shows some common metric prefixes (a similar table is also discussed in the scientific notation module).
Table 2. Metric prefixes
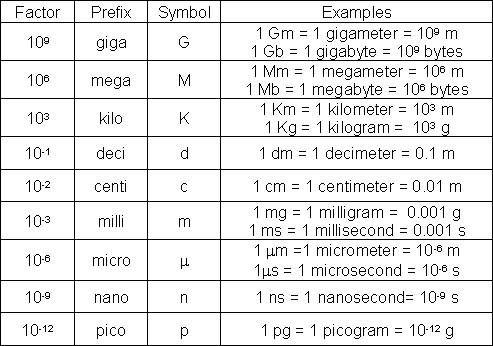
Image: www.webassign.net/question_assets/ncsugenchem102labv1/
The following link to Math Is Fun - Maths Resources discusses metric numbers and the metric system. Please read through the explanations and information provided and answer the practice questions which can be found by clicking on each of the question numbers located towards the end of the web page. We recommend that you complete questions 4 to 10.
Perhaps the most common metric conversions are between mm, cm, m and km.
1 cm = 10 mm
1 m = 100 cm = 1000 mm
1 km = 1000 m = 100 000 cm = 1 000 000 mm
kilo means 1000, so 1 km = 1000 m
centi means ![]() th, so 1 cm =
th, so 1 cm = ![]() th m
th m
milli means ![]() th, so 1 mm =
th, so 1 mm = ![]() th m
th m
Metric prefixes are consistent across all units of measurement. For example, just as one milligram is one thousandth of a gram and one microgram is one millionth of a gram, one millimole is one thousandth of a mole and a micromole is one millionth of a mole. The diagram below shows the relationship between grams, milligrams and micrograms.
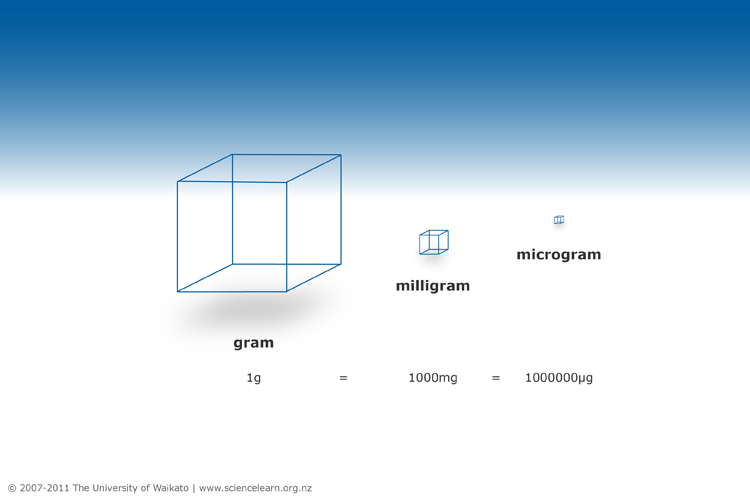
Image: https://www.sciencelearn.org.nz/images/570-the-relationship-between-a-gram-milligram-and-microgram
Many of the calculations in chemistry involve mass and volume. The tables below give some common volume and mass measures.
10 millilitres (ml) = | 1 centilitre (cl) | |
10 centilitres = | 1 decilitre (dl) | = 100 millilitres |
10 decilitres = | 1 litre (l) | = 1,000 millilitres |
10 litres = | 1 dekalitre (dal) | |
10 dekalitres = | 1 hectolitre (hl) | = 100 litres |
10 hectolitres = | 1 kilolitre (kl) | = 1,000 litres |
10 milligrams (mg) = | 1 centigram (cg) | |
10 centigrams = | 1 decigram (dg) | = 100 milligrams |
10 decigrams = | 1 gram (g) | = 1,000 milligrams |
10 grams = | 1 dekagram (dag) | |
10 dekagrams = | 1 hectogram (hg) | = 100 grams |
10 hectograms = | 1 kilogram (kg) | = 1,000 grams |
1,000 kilograms = | 1 metric ton (t) |
Tables from: www.infoplease.com/ipa/A0001659.html
Energy requirements of the body
The following information was adapted from the following source:
http://www.nrv.gov.au/dietary-energy
Energy is required in the body for metabolic processes, physiological functions, muscular activity, heat production, growth and synthesis of new tissues. It is released from food components by oxidation.
The main sources of energy are carbohydrates, proteins, fats and, to a lesser degree, alcohol.
Energy requirements vary with age, gender, body size and activity.
The unit of energy is the kilojoule (kJ) or megajoule (1 MJ = 1,000 kJ)
4.18 kilojoules are equal to 1 kilocalorie.
Metabolic rates
This section was adapted from the following source:
The amount of energy needed to keep the body functioning in a resting, awake and fasting state in a comfortably warm environment is called the resting metabolic rate (RMR) sometimes called the basal metabolic rate (BMR)
The total metabolic rate is the amount of energy used by the body in carrying out all of its activities so it is higher than the RMR.
It is often expressed in kilojoules or kilocalories per day.
The factors that determine the total metabolic rate are:
Equation for calculating RMR
A relatively simple way of determining resting metabolic rate is to use an equation known as the Mifflin equation. It is based on body mass (w), height (h), age (a) and sex.
Although it does not take ethnicity or amount of fat tissue to lean tissue into account, it is still a good predictive measure.
The units of the resulting RMR calculated using the following formula, is the kilocalorie. To convert kilocalories to kilojoules you need to multiply by 4.18 (recall the conversion factor mentioned previously).
Example
A teenage boy of body mass 60kg, height 165cm, age 14 years:
RMR | = (10 x 60) + (6.25 x 165) - (5 x 14) + 5 |
= 600 + 1,031.25 - 70 + 5 | |
= 1,566.25 kilocalories x 4.18 to convert to kilojoules | |
= 6,546.9 kJ per day |
A teenage girl of body mass 50 kg, height 163 cm, age 14 years:
RMR | = (10 x 50) + (6.25 x 163) - (5 x 14) - 161 |
= 1,287.75 kilocalories x 4.18 to convert to kilojoules | |
= 5,382.8 kJ per day |
A calculation involving metric units:
A person's body mass index (BMI) can be calculated as a guide to whether their
weight is within a recommended range for their height. The following table provides some broad BMI categories for adults in Australia

Image: www.aph.gov.au/binaries/library/intguide/sp/obesity-1.jpg
The formula for BMI is:
![]()
Where w = the person's weight in kilograms
h = the person's height in metres
Example 1
Calculate the BMI of a person with a height of 170 centimetres and a weight of 65 kilograms.
First change the height to metres 170 cm = 1.7 m (dividing by 100)
![]()
![]()
BMI = 22.49
On most scientific calculators enter 65 ÷ 1.7 followed by the 'squared' key which is likely to be labelled x2 on your calculator's keypad. There are also a range of online BMI calculators.
Example 2
Most of the industrial world uses metric units (base 10 units), but imperial measures are also still widely used. In the USA, imperial measurements are nearly always used. In the USA feet and inches are often still used to measure height, and stones and pounds to measure weight.
The following example was adapted from the following online source
The following information may assist you to answer the questions below:
1 kg = 2.2046 lb (pounds)
1 inch = 2.54 cm
12 inches = 1 foot
14 pounds = 1 stone
According to a source Johnny Depp is 5 feet 10 inches and weighs 155 pounds (lb)
Convert these imperial measurements to metric measurements and calculate Johnny's BMI
Solution: First convert the weight and height measurements from imperial to metric.
5 feet 10 inches = 70 inches (given that 1 foot is equal to 12 inches so 5 feet equals 60 inches)
70 inches = (70 x 2.54) kg (given then 1 inch is equal to 2.54 cm)
70 inches = 177.8 cm = 1.778 m
To convert pounds to kg is a little trickier given that 1 kg = 2.2046 pound
This means that 1 pound = ![]() = 0.4536 kg
(to 4 decimal places)
= 0.4536 kg
(to 4 decimal places)
Therefore 155 lb =( 155 x 0.4536) kg = 70.31 kg
![]()
![]()
BMI = 22.24
Complete the following unit conversions:
1. There are ___ in 2000 mm?
A 200m
B 2m
C 0.002m
D 0.02m
2. There are ____ in 0.05 ml?
A 0.00005 L
B 5 L
C 50 L
D 0.0005 L
3. 30 mg is the same mass as:
A 300 decigrams
B 0.3 grams
C 0.0003 kg
D 0.03 g
4. 30 microlitres is the same as:
A 30000000 L
B 30000 decilitres
C 0.000003 L
D 0.03 ml
5. 20 grams is the same as:
A 2000 mg
B 20000 mg
C 200000 mg
D 200 mg
6. 4 g is equal to:
A 0.04 mg
B 0.4 mg
C 400 mg
D 4000 mg
7. There are ____ in 12.0 ml?
A 12000 L
B 120 L
C 0.012 L
D 0.12 L
Click here to check your answers
For further practice with metric volume and mass, the following web links provide an overview of the important ideas associated with the quantities of mass and volume as well as some practice questions (you will find links to the questions towards the end of each of the following webpages).
These links to the Math Is Fun - Maths Resources explains some important ideas about metric volume and metric mass including the relationship between litres and cubic metres.
Click here to check your answers.
The relationship between density, mass and volume is demonstrated in the following brief YouTube video. Note that we are specifically dealing with mass density.
In the video clip we saw that the block of wood and the sponge had the same mass, but the volume of the block of wood was less that the volume of the sponge.
In other words the density of the block of wood is greater than the density of the sponge.
Similarly, if we adjusted the dimensions of the block of wood and the sponge so that they both had the same volume, then the mass of the block of wood would be greater than the mass of the sponge.
In summary, the density, or more precisely, the volumetric mass density, of a substance is its mass per unit volume. The symbol for density is ρ (the lower case Greek letter rho). Mathematically, density is defined as mass divided by volume:
![]()
Where, ρ is the density, m is the mass, and V is the volume.
An alternative symbol for density that we often see is D, such that
![]()
Where, D is the density, m is the mass and V is the volume. We will use this notation in this module.
You may find the diagram below helpful in rearranging the equation, ![]() to make either m the subject or V the subject.
to make either m the subject or V the subject.
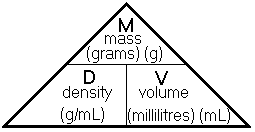
Image: //1.bp.blogspot.com/-ngu-O3qXiEY/UqYm3SpsGDI/AAAAAAAAC_Q/mgVPvOmSzmQ/s400/density.gif
Thus we have,
m = D x V that is mass = Density x Volume
and
![]() that is Volume = mass ÷ Density
that is Volume = mass ÷ Density
We know that the density is equal to mass per unit volume.
![]()
More specifically let us consider the units of mass, for example grams (g) and the units of volume, for example millilitres (mL), it follows that the unit of density, in the case of this example, is grams per millilitre (g/mL). We would expect that the units such as litres (L) and mL(millilitres) are appropriate for measuring the volume of liquids, but for measuring the volume of a solid we would use units such as cm3 (cubic centimetres) or m3 (cubic metres). The SI unit for mass density is kg / m3 (SI units are discussed in the Scientific Notation module).
Note also that:
1 cm3 = 1 mL
1 m3 = 1 L
Click here to check your answers
The following You Tube clip provides some further examples involving calculating density.
We recommend that you pause the video after each problem is presented, do the calculation yourself and then resume the video to see the presenter's solution. You will notice that the presenter particularly attends to the units of density in each of the problems.
Example
Watch how the presenter uses ![]() as a conversion factor to ensure that the units of density will be in g/mL. Compare this to the method of solution used in practice examples 1, 2 and 3 where the volume has been converted to mL before substitution into the equation.
as a conversion factor to ensure that the units of density will be in g/mL. Compare this to the method of solution used in practice examples 1, 2 and 3 where the volume has been converted to mL before substitution into the equation.
Percentage composition by mass of compounds problems
The following example involves finding the percentage composition by mass of a substance.
Worked example
A 4 g sugar cube (Sucrose: C12H22O11)
is dissolved in a 350 ml teacup of water of temperature 80°C. What is the
percentage composition by mass of the sugar solution?
Given: Density of water at 80 °C = 0.975 g/ml
Solution:
First record all the information we have from the question
mass solute= 4 g (remember the solute is the substance that we dissolve in the solvent and in this case it is the sugar (sucrose)).
mass solvent= the mass of the water.
Now here is where we need to use the information we were given about the density of water at 80°C (i.e., 0.975 g/ml). We also know the volume of water is 350ml. Hence:
![]()
m = D x V
m = 0.975 g/ml x 350 ml
m = 341.25 g
Hence the total mass of the solution is:
msolution = msolute +
msolvent
msolution = 4 g + 341.25 g
msolution = 345.25 g
To determine the percentage composition by mass of the sugar solution, we may reason as follows:
Remember that mass of solute is the mass of the sugar and the mass of the solution is the total mass of the solute and the solvent. Let the percentage composition by mass of sugar be x
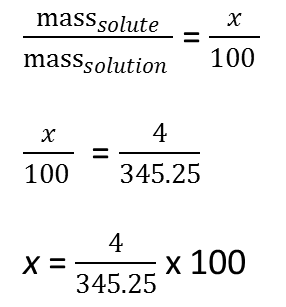
Note: in mass solute / mass solution = x /100, we have set up a proportion.
Percentage composition by mass of sugar = 1.16%
The answer is rounded to 2 decimal places.
Alternatively you could approach
the calculation in the following way:percent composition = (msolute /
msolution) x 100
percent composition = (4 g / 345.25 g) x 100
percent composition = (0.0116) x 100
percent composition = 1.16%
On your calculator you would
enter 4 ÷ 345.25 x 10
Before we look at how particular
calculations work, we need to be familiar with some common chemistry terms in the next tab (solute, solvent and solution)
A solution is a homogeneous (uniform) mixture of two or more substances.
While we may often think of solutions as being in a liquid form, a solution may exist in any phase (e.g., solid, liquid, gas).
A solvent is the component of a solution that is present in the greatest amount and it is the substance in which the solute is dissolved. For example, the solvent for seawater is water.
The substance that is dissolved in a solution is the solute. For example, the solute in seawater is salt.
Example
In the figure below where the solute is sugar, the solvent is water and the resulting solution is sugar dissolved in water, and the solution is the same composition throughout (in this case the same "sweetness" throughout). We therefore say that the solution is homogenous, or uniform in composition. Although we may normally think of a solute as a solid that is added to a solvent, it is important to note that a solute does not necessarily have to be a solid.
The mole is a unit of measurement used in chemistry to express amounts of a chemical substance. The mole is a base unit of the International System of Units (SI). The following video examines the mole.
Concentration
The concentration of a solution is the amount of a substance per defined space. Concentration usually is expressed in terms of mass or amount per unit volume (e.g., g/cm3, kg/L).
Example
If we put 1 gram of salt (e.g., sodium chloride) in a beaker, that it is an amount (i.e., 1 gram of salt) but if we bring the volume up to 1 litre with water then the concentration of our resulting solution would be 1 g/L (i.e., 1 gram per litre). We would say that a solution with a concentration of 2g/ L is more concentrated than a solution that has a concentration of 1g/L.
In chemistry, the concentration of a solution is commonly expressed as moles per litre.
In this module we will focus on the following units of concentration: grams per litre (g/L), moles per litre (mol/L), and millimoles per litre (mmol/L).
The concentration of a chemical solution that you will most likely use in your health science studies is:
Molarity, M = moles solute / litre of solution
You will also come across units of concentration such as micromoles per litre (µmol/L) and millimoles per litre (mmol/L).
Note: however, that there are several other ways of expressing concentration that are not dealt with in this module.
Dilution is the process of adding a solvent to a solution to lower its concentration. The solvent that is used to lower the concentration of a stock solution is called the diluent. The stock solution is a concentrated solution that will be diluted to some lower concentrated for actual use.
Dilution Calculations
The formula for calculating a dilution is based on the concept of proportion.
C1V1 = C2V2 where,
C1 = initial concentration of the solution
V1 = initial volume of the solution
C2 =final concentration of the solution
V2 =final volume of the solution
Worked example 1
If we have a 5M solution of salt (NaCl) and we want to prepare 100 mls of a 0.5M solution. What is the initial volume of the 5M solution? Recall that "M" means moles/litre.
C1V1 = C2V2
C1 = 5M (or 5 m/L)
V1= ? this is our unknown quantity
C2=0.5M
V1= 100 mL
C1V1 = C2V2
5 x V1 = 0.5 x 100
![]()
V1 = 10 mL
Therefore the volume of the 5M solution required is 10 mL
Worked example 2
The following examples have been adapted from a publication by Dr Dale Kunde that was designed to assist students with their understanding the calculations associated with dilutions.
I want to prepare 10 mL of a 1.5 mmol/L solution of glucose from a stock mmol/L solution of glucose. How much stock solution do I need to dilute to 10 mL to get my 1.5 mmol/L solution of glucose? Recall that a stock solution is a concentrated solution that will be diluted to some lower concentrated for actual use.
C1V1 = C2V2
Where C1 = 60 mmol/L
V1 = ? we need to work this out
C2 = 1.5 mmol/L
V2 = 10 mL
60 x V1 = 1.5 x 10
![]()
V1 = 0.25 mL
Therefore you need to take 0.25 mL of the stock solution and dilute to 10 mL with the diluent (i.e., the diluting agent) and you have a 1.5 mmol/L solution of glucose.
Now to get this 10 mL you need to add 9.75 mL of diluent to your 0.25 mL of stock solution.
In other words, we calculated the volume of the stock solution that we would need to dilute to get a total volume of 10 ml. So to get the 10 ml we use the 0.25 mL of stock solution and add 9.75 mL of diluent to make up the 10 mL.
Worked example 3
I diluted 1 mL of my stock creatinine solution that has a concentration of 950 µmol/L with 9 mL of diluent. What is the creatinine concentration in my new solution?
C1V1 = C2V2
Where C1 = 950 µmol/L
V1 = 1 mL
V2 = ? We need to work this out
V2 = 10 mL
C1V1 = C2V2
950 x 1 = C2 x 10
950 = C2 x 10
![]()
C2= 95 µmol/L
Therefore the final concentration is 95 µmol/L
Dilution factors
If I dilute something![]() this is the
same as saying I have diluted 1 volume of the initial solution (V1) to
a final volume of 10 volumes of diluent (V2).
this is the
same as saying I have diluted 1 volume of the initial solution (V1) to
a final volume of 10 volumes of diluent (V2).
This means that our dilution factor is![]()
It does not matter what the actual volumes are, it is the ratio that is important.
Example
10 µL stock + 90 µL diluent. Remember that your initial volume V1 is 10 µ L and the final volume V2 is 100 µ L so this is why you need to add 10 µL of the stock solution to 90 µL of the diluent.
Here are some other examples of ![]() dilutions:
dilutions:
1 µL stock + 9 µL diluent
1000 L stock + 9000 L diluent
Worked example 4
What is the dilution factor if I take 200 µL of serum and add 800 µL of isotonic saline?
Answer: The dilution factor ![]()
Note that the isotonic saline is the diluent. It is important not to confuse this and think that the final volume is 800 µL.
Worked example 5
We can
also deal with dilution factors involving concentrations. For example, if we
start out with 20 g/L solution and end up with 1 g/L solution then my dilution
factor is ![]() . To
explain why this is the case consider our proportional relationship C1V1 = C2V2
. To
explain why this is the case consider our proportional relationship C1V1 = C2V2
Now if we look at this relationship in a slightly different (but equivalent) way we see that
![]()
In other words the ratio of the initial volume to the final volume is equal to the ratio of the final concentration to the initial concentration. Note that the actual volumes are not known, it is the ratio that is important.
Click here to check your answers
Click on the link below to take the online self-assessed quiz.
There are 10 mathematics questions on the Quiz and they are about the information in this module.
To pass this quiz, you will need to get a mark of 80%. Feedback will be provided for both correct and incorrect answers at the end of the Quiz. If you answer questions incorrectly, then it is strongly recommended that you review the sections of the modules to review those topics. You will be able to re-take the quiz if needed.
Make sure to enter your name and email address in the quiz so your results can be mailed to you for your records. You may need to show your results to your university.
Authorised by the Director, Centre for University Pathways and Partnership
2 May, 2018
Future Students | International Students | Postgraduate Students | Current Students
© University of Tasmania, Australia ABN 30 764 374 782 CRICOS Provider Code 00586B
Copyright | Privacy | Disclaimer | Web Accessibility | Site Feedback | Info line 13 8827 (13 UTAS)