UTAS Home › › Mathematics Pathways › Pathways to Health Science › Module 7: Scientific Notation
Click on the link below to take the Pre-Test for Module 7. The Pre-Test is optional but we recommend taking it to test your knowledge of Scientific Notation. There are only 5 questions and it will only take about 10 minutes to complete.
If you receive 80% or greater on the Pre-Test, you have a good knowledge of Scientific Notation and can move on to the next module or review the materials in module 7.
If you receive less than an 80%, work your way through the module and then take the quiz at the end to test your knowledge.
In the context of physical science, physical quantities are often referred to as variables. A physical quantity, or variable, is anything to which we can assign a numerical value via some kind of measurement (e.g., length, mass, time).
Example
The height of a building, the diameter of a proton, the distance from the Earth to the Moon, the thickness of a piece of paper all involve a measurement of a length, L, of some kind.
These things are all measured in different ways and have their values assigned using different units, but they all share the common feature of being a kind of length.
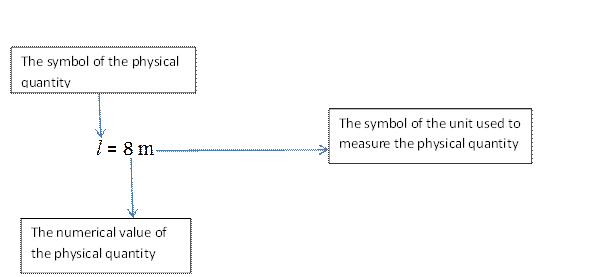
Suppose that we measure the length of a room and find that it is 8 metres long. The length of the room is the physical quantity (the symbol for length is l, the 8 is the numerical value of this length and the m is the symbol of the unit used to measure this length, as illustrated in the diagram below.
The following video provides a clear explanation of some the profound and fundamental ideas.
Practice Task 1
For each of the following indicate
Click here to check your answer
This system is a globally agreed system of units, comprising two classes of units: base units and derived units. There are seven base units, and these are listed below:
Kilograms, metres and seconds are familiar to us be as being fundamental quantities, whereas the others are less obviously fundamental. What is important, however, is that ANY physical quantity can be measured by one, or a combination of the base units.
Other SI units of measurements, are shown below in tables 1 and 2. While it is not necessary for you to memorise all these units, you may notice the underlying simplicity in that each of these derived units are combinations of one or more of the seven base units.
| Table 1 |
|
|
Table 2 provides a list of special SI units. These special SI units are also derived from the original base units but because they are the result of more complex derivations, for convenience and simplicity they have been given their own special name.
| Table 2. SI derived units with special names |
| Physical Quantity | Quantity symbol | SI Unit | Unit Symbol | Expression in SI base units | Alternative expressions |
| frequency | v,f | hertz | Hz | s-1 | - |
| force | F | newton | N | kg m s-2 | J m-1 |
| pressure | P | pascal | Pa | kg m-1 s-2 | N m-2 |
| energy (all forms) | E, U, V, W, etc | joule | J | kg m2 s-2 | N m = C V = VA s |
| power | P | watt | W | kg m2 s-3 | J s-1 = VA |
| electric charge | Q | coulomb | C | A s | - |
| electric potential difference | E, Φ, η, ψ, etc | volt | V | kg m2 s-3 A-1 | J A-1 s-1 = J C-1 |
| electrical resistance | R | ohm | Ω |
kg m2 s-3 A-2 | VA-1 |
| electrical conductance | G | siemens | S | A2 s3 kg-1 m-2 | VA-1 = Ω-1 |
| activity (of a radionuclide) | A | becquerel | Bq | s-1 | - |
| absorbed dose | D | gray | Gy | m2 s-2 | J kg-1 |
| dose equivalent | H | sievert | Sv | m2 s-2 | J kg-1 |
| Celsius temperature | t | degree Celsius | °C |
K | - |
Table 2 was recreated from, http://www.gordonengland.co.uk/conversion/siderived1.htm
There are some other units, and you will most likely be familiar with some of them, that are not SI units, but they are in continuous everyday use and are accepted for use along with the International System.
Example
The traditional units of time (e.g., hours, minutes) and angle (degrees) are non SI units. The table below provides a list of some non-SI units (note that this list is by no means an exhaustive list of non-SI units, there are others).
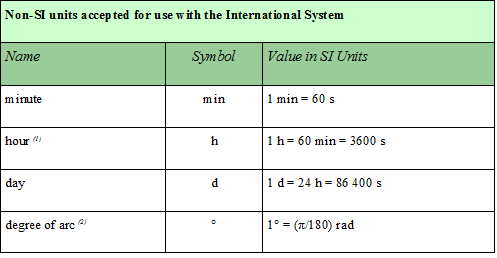
Table from http://physics.nist.gov/cuu/Units/outside.html
Imperial Units
There are some imperial units still in use particularly in the USA
Example
gallons (1 gallon =3.7854 litres) and miles (1 mile = 1609.3 metres)
Use the information from the tables presented in Big Idea 1, along with the seven SI base units to answer the following:
Click here to check your answers.
Sometimes the units we use to measure a particular quantity are too large or too small to represent a quantity. Therefore, we use standard prefixes to modify the unit. Some examples of such modifications are shown below:
The following table provides a list of some common standard metric prefixes. Note that there are other metric prefixes that stand for larger and smaller orders of magnitude.
Example
The largest accepted metric prefix is the yotta which is 1 000 000 000 000 000 000 000 000 or 1024
The smallest accepted metric prefix is the yocto which is 0.000000000000000000000001 or 10-24
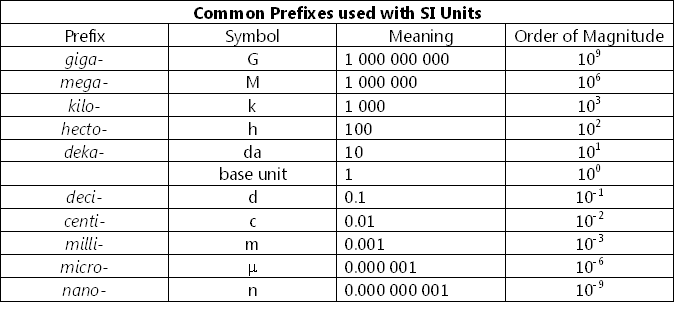
http://sciencepedagogics.pbworks.com/f/1311886367/PREFIXES.PNG
Let us examine the symmetrical pattern shown in the third column of the above table. Starting from the top of the column (i.e., giga: 1000 000 000) you will notice that as you move down the column, each number is 10 times smaller than the one above it.
Similarly, if you start at the bottom of the column (i.e., nano: 0.000 000 001), and move up, each number is 10 times bigger than the one below it. Numbers such as 106 and 10-6 are examples of “powers of ten”. The illustration below shows the structure of this notation, where 10 is the base and the number in the superscript is called the exponent, or power (or it can also be called the index). We can also call this index notation, or exponential notation.

Image: Index notation terminology adapted from Index Notation - Powers of 10
In order to investigate powers of 10 in more depth, take a look at table 5 which shows the different ways of expressing powers of ten.

Image saved from: https://share.ehs.uen.org/node/3146
Use the following table to answer these practice questions

Click here to check your answers
Before we discuss scientific notation in some detail, take a look at the following youtube clip as it highlights how scientific notation makes it so much more convenient to represent very, very large and very, very small numbers.
The range of objects and phenomena studied in our world is immense.
From the tiny size of a sub-nuclear particle to the astronomically large distance to the most distant object seen in the known universe, there are numbers so large and others so small that they challenge our imagination.
Example
The mass of the sun is
1 989 100 000 000 000 000 000 000 000 000 kg
The mass of a virus particle is about
0.00000000000001 grams.
Once numbers become very large, a more efficient system of notation is required. The system used is called scientific notation.
The following video explains how to express numbers in scientific notation.
Now that you have watched the video, please complete the 5 practice questions on scientific notation by clicking on the following link to khan academy.
Note: that standard form is just the ordinary decimal form of a number. For example, 3.5 x 105 written in standard form is 350 000.
As you may have observed, scientific notation specifies a value as a number between 1 and 10 (called the mantissa below) multiplied by some power of ten, as shown below:
mantissa×10exponent
Example
The value 6.02 x 1023
6.02 is the mantissa and the 23 is the exponent (also called the power).
If the scientific form of a number has a negative exponent (e.g., 7.6 x 10-7), then it is a very small number.
We need to be careful here though because 7.6 x 10-7 is still a positive number.
That is, 7.6 x 10-7 is located to the right of zero on the number line, between zero and positive one (albeit closer to zero than one). On the other hand, think about where we would place the number -7.6 x 10-7 on the number line? This number is negative, so it would lie to the left of zero on the number line, somewhere between zero and negative one (albeit closer to 0 than -1). It is important to understand that a negative power (exponent) and a negative number mean two very different things and the distinction between them is summarised in the diagram below.

The following video explains the purpose of using significant figures and how this relates to the concepts of Precision and Accuracy. The presenter also provides a clearly explains the rules for determining the number of significant figures in a measurement.
Now that you have watched the video, please click on the Khan academy link below and complete some practice examples, and then complete the further practice examples below.
1. Write each of the following numbers in standard form. For example 2.6 x 103 is equivalent to 2600 in standard form.
-3.6 x 10-4
3.6 x 10-4
3.6 x 104
-3.6 x 104
2. For each of your responses to question 1 state:
3. Write 3.6 x 〖10〗^12 in standard form.
4. Write 0.000 000 000 043 6 in scientific notation.
5. Convert 0.000 000 005 78 to scientific notation.
6. Convert 93,000,000 to scientific notation.
Click here to check your answers
Rounding a numerical value means replacing it by another value that is approximately equal but has a shorter, or simpler representation. The following examples explain how to round off numerical values to a particular number of significant figures.
Example 1
Round 92.810576 to 1 significant figure
Answer: 9 is the first non-zero digit, which means that it is the first significant figure. In 92.810576 the second figure 2,is less than 5, so we round down the number.
When we round off 92.810576 to 1 significant figure The answer is 90
Example 2
Round 92.810576 to 3 significant figures
Answer: In 92.810576, 928 are the first three significant figures. The next figure 1 is less than 5, so we round down the number.
When we round off 92.810576 to 3 significant figures the answer is 92.8
Example 3
Round 0.0046753 to 1 significant figure.
Answer: In 0.0046753, 4 is the first significant figure. The leading zeros are not significant, but they are used to keep other figures in their correct places .In the above number the figure to the right of 4, is 6 which is more than 5, so we round up the number.
When we round off 0.0046753 to 1 significant figure the answer is 0.005
Example 4
Answer: In 0.0046753, 4 is the first significant figure. The leading zeros are not significant, but they are used to keep other figures in their correct places. In the above number, 5 is the 4th significant figure. The figure to the right of 5 is 3 which is less than 5, so we round down the number.
When we round off 0.0046753 to 4 significant figures the answer is 0.004675.
For some further information and clarification on significant figures you may wish to read through the information found in the following weblink: Significant Figures- Rules and you will also find some worked examples that use the rules for determining the number of significant figures in a number.
1. How many significant figures are in the number 720?
A) 1
B) 2
C) 3
2. How many significant figures are in the number 8.4 x 103
3. How many significant figures are in the number 20.003?A) 2
B) 3
C) 4
D) 5
A) 2
B) 3
C) 4
D) 5
4. How many significant figures are in the number 0.00300?
Click here to check your answersA) 1
B) 2
C) 3
D) 5
Make sure that the number in scientific notation is put into your calculator correctly.
You are encouraged to read the instructions for your particular calculator. In general, for a scientific calculator you would use the EXP or 10x button. For example, suppose you want to enter the number 2.3x10-4 you would enter it as follows:
EXP(-) 4 enter
or
2.3 10x4
Make sure that you use the “negative” key (e.g., (-)) and not the minus operator button when you are entering negative exponents.
Practice Task 3
Complete the following calculations using your scientific calculator. You do not need to enter the brackets on your calculator.
(4.7 x 1010) + (35.6 x 108) =
(3.72 x 10-4) – (8.21 x 10-2) =
(-1.87 x 103) + (2.1 x 104) =
Click here to check your answers
Click on the link below to take the online self-assessed quiz.
There are 10 mathematics questions on the Quiz and they are about the information in this module.
To pass this quiz, you will need to get a mark of 80%. Feedback will be provided for both correct and incorrect answers at the end of the Quiz. If you answer questions incorrectly, then it is strongly recommended that you review the sections of the modules to review those topics. You will be able to re-take the quiz if needed.
Make sure to enter your name and email address in the quiz so your results can be mailed to you for your records. You may need to show your results to your university.
Authorised by the Director, Centre for University Pathways and Partnership
2 May, 2018
Future Students | International Students | Postgraduate Students | Current Students
© University of Tasmania, Australia ABN 30 764 374 782 CRICOS Provider Code 00586B
Copyright | Privacy | Disclaimer | Web Accessibility | Site Feedback | Info line 13 8827 (13 UTAS)